Home
Guides
Important Contamination Control Strategies for Pharma and Biotech Manufacturing
Your organization operates in environments where even the tiniest particle, microbe, or deviation can have serious consequences. High-value therapies depend on well-controlled cleanrooms that protect product quality, patient safety, and regulatory compliance. If contamination enters the process, it can compromise entire batches, halt production, or trigger regulatory penalties. For this reason, every facility must maintain a strong contamination control program—a structured, holistic system that manages contamination risks throughout manufacturing.

Regulators today expect organizations to implement integrated, risk-based contamination control strategies that go far beyond basic housekeeping. EU GMP Annex 1 emphasizes contamination control as a lifecycle discipline. FDA, Health Canada, and EMA authorities increasingly expect facilities to show how design, behaviors, airflow, cleaning, gowning, and environmental monitoring work together to reduce contamination risk. In modern pharma and biotech environments, contamination control is not simply about reacting to problems as they arise; it is about preventing them through structured, scientifically validated systems.
This article provides a detailed look at the most critical contamination control strategies used across pharma, biotech, radiopharmaceutical, and ATMP manufacturing. It outlines the sources of contamination, how facilities can prevent it, and how to maintain a compliant, audit-ready state at all times. Whether you operate a single graded room or a multi-suite manufacturing campus, these foundational practices strengthen contamination prevention in pharma and support safe, uninterrupted production.
Understanding the Sources and Risks of Contamination
This includes residues from raw materials, cleaning chemicals, detergents, or disinfectants. Chemical contamination is particularly critical in facilities that produce potent or allergenic compounds. Cross-contamination between products—particularly high-potency APIs, hormones, or biologics—can lead to severe regulatory findings and patient risks.
Different pharma and biotech environments face unique contamination risks:
- Bioreactors may face microbial ingress from connections or gaskets.
- Radiopharma environments often include open handling of materials that cannot be reworked.
- Compounding pharmacies handle potent ingredients with high sensitivity.
- ATMP and cell therapy facilities face high microbial sensitivity due to live cell products.
A successful contamination control program begins with identifying and ranking these risks.
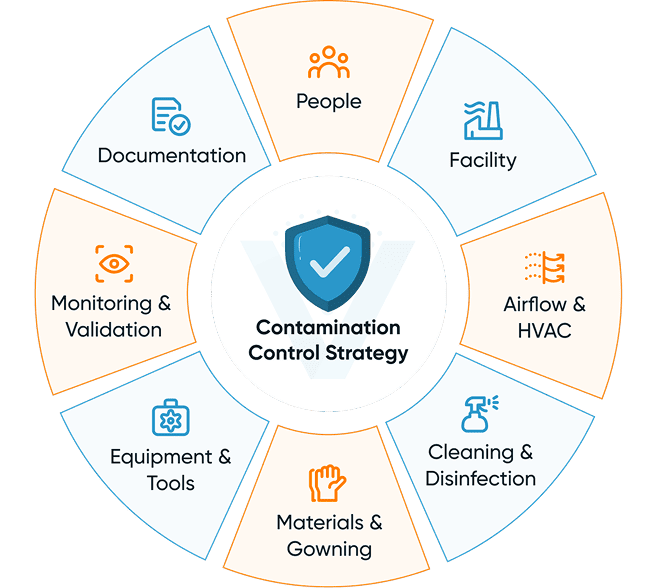
Building a Modern Contamination Control Strategy (CCS)
A Contamination Control Strategy (CCS) is now a regulatory expectation, especially under Annex 1. It demonstrates how all contamination risks are identified, assessed, controlled, and reviewed.
Regulators look for a strategy that:
- Identifies contamination risks across the entire process.
- Justifies how each risk is controlled.
- Demonstrates integration between design, cleaning, gowning, and monitoring.
- Shows data-driven decision-making.
- Is reviewed periodically and improved continuously.
FDA guidance emphasizes scientific rationale, repeatability, and clear traceability. Annex 1 expects facilities to “establish, document, implement, and maintain” a CCS that unifies all contamination controls.
A comprehensive CCS includes:
- Risk assessments for processes, utilities, and equipment.
- Environmental and personnel monitoring design.
- Cleaning, disinfection, and decontamination strategies.
- Gowning and behavioral expectations.
- Change control and deviation management.
- Validation requirements for cleaning and disinfectants.
Each component must align with quality systems and support defensible decision-making.
Quality plays a central role in contamination control by overseeing:
- SOP review and approval.
- Deviation investigations.
- CAPA integration.
- Training oversight.
- Change control for facility or procedure modifications.
A CCS must be fluid, updated as processes evolve, and supported by cross-functional ownership.
Risk-based facility design
- Smooth, seamless surfaces without ledges.
- Rounded transitions and easy-clean corners.
- Minimal fixtures or penetrations.
- Epoxy, PVC, or polymer flooring suitable for disinfectants.
- Controlled temperature and humidity.
HVAC and HEPA filtration
- HEPA-filtered supply air.
- Pressure differentials between rooms.
- Controlled air changes per hour.
- Unidirectional airflow for critical zones.
- Routine HEPA integrity testing.
Personnel and material flow
Cross-flows are a significant contamination risk.
Controls should include:
- Unidirectional personnel movement.
- Dedicated gowning rooms per cleanliness level.
- Material airlocks with transfer disinfection.
- Pass-throughs for tools or components.
- Electronic interlocks on cleanroom doors.
Designing flows that reduce backtracking and cross-traffic helps reduce overall contamination.
Commissioning and recommissioning
Cleanroom Zoning and Segregation
Zoning prevents higher-risk processes from affecting lower-risk areas and keeps incompatible activities physically separated.
Zoning principles
Cleanrooms should be divided into:
- Classified process areas.
- Support rooms.
- Intermediate spaces (airlocks, gowning).
- Material preparation rooms.
Each zone must reflect the contamination risk and have defined behaviors and cleaning expectations.
Segregation controls
Segregation prevents cross-contamination by using:
- Physical walls or barriers.
- Dedicated tools and cleaning materials.
- Equipment segregation.
- Campaign-based manufacturing.
Material transfer
Materials introduce contamination every time they cross a boundary. Effective controls include:
- Wipe-down with approved disinfectants.
- Double-bagging and staged removal.
- Pass-through chambers.
- Unidirectional movement through airlocks.
Segregation is one of the most impactful strategies in a contamination control program because it prevents contamination spread at its source.

Validated Cleaning and Disinfection Programs
Cleaning and disinfection are central to removing contamination from surfaces, equipment, and rooms.
SOP structure and clarity
Cleaning SOPs must define:
- Scope and responsibilities.
- Tools and materials.
- Step-by-step cleaning sequence.
- Disinfectant instructions and contact times.
- Documentation steps.
Diagrams, labeled images, and tool diagrams reduce operator variability.
Choosing the right disinfectants
A strong disinfectant program uses:
- Broad-spectrum disinfectants.
- Sporicidal agents.
- Residue management strategies.
- Compatibility evaluations.
Disinfectants must match the facility’s environmental flora, especially in sterile manufacturing.
Disinfectant rotation
Rotation prevents microbial adaptation. Regulators expect rational, scientifically supported rotation that includes:
- Alternating disinfectant classes.
- Periodic sporicidal exposure.
- Use of detergents to remove residue.
Rotation should be facility-specific and justified in validation reports.
Cleaning validation
Validation ensures cleaning reduces contamination to acceptable levels. A typical validation includes:
- Acceptance criteria based on risk and classification.
- Worst-case surface and location selection.
- Contact plate or swab sampling.
- Three consecutive successful cycles.
- BI/CI placement for sporicidal validation.
- Detailed final validation reports.
Cleaning validation is one of the strongest indicators of a mature contamination control system.
Daily vs periodic cleaning

Personnel Training, Gowning, and Behavior Control
Human operators are the primary source of contamination. Strong contamination control depends on disciplined personnel practices.
Training and competency
Training must cover:
- GMP fundamentals.
- Aseptic technique.
- Cleanroom behavior.
- Cleaning steps and disinfectant use.
- Emergency procedures.
Competency assessments and periodic retraining ensure consistency.
Gowning systems
Gowning garments act as barriers to contamination. Strong systems include:
- Validated gowning sequences.
- Garment integrity checks.
- Proper glove management.
- Defined change frequencies.
Gowning qualification should be documented and renewed periodically.
Behavior control
- Slow, deliberate movements
- Minimal talking
- Avoiding unnecessary touching
- Mindful tool handling
Behavioral drift is a major cause of contamination; auditing operator practice is essential.
Environmental Monitoring (EM) as a Verification Tool
EM confirms that contamination controls are effective. It provides data that supports decision-making and regulatory compliance.
EM program design
A robust EM program includes:
- Non-viable particulate monitoring.
- Viable air sampling.
- Surface sampling (contact plates, swabs).
- Personnel monitoring.
- Environmental trending.
- Defined action and alert limits.
Monitoring should reflect worst-case conditions.
Trending and interpretation
Trending is one of the most useful contamination control tools. Trends identify:
- Seasonal variations.
- Operator-specific issues.
- Process-related spikes.
- Room deterioration.
- Cleaning program weaknesses.
Handling EM excursions
Excursions require:
- Investigation.
- Root cause analysis.
- CAPA.
- Potential recleaning.
- Requalification if needed.
EM data should inform updates to cleaning frequency, disinfectant rotation, gowning practices, or airflow adjustments.
Decontamination Technologies for High-Risk Events
Decontamination goes beyond daily cleaning to restore sterility or reset cleanrooms after contamination events.
When decontamination is required
Typical triggers include:
- Construction or maintenance.
- HVAC failures.
- Mold detection.
- Spills.
- Shutdown/restart cycles.
- EM excursions.
Hydrogen peroxide-based systems
These systems provide broad-spectrum, residue-free decontamination. Technologies include:
- Vaporized hydrogen peroxide (VHP/HPV).
- Hydrogen peroxide fogging.
- Ionized hydrogen peroxide (iHP®).
Each technology offers different depths of penetration, cycle times, and material compatibility.
Biological indicator validation
BI placement ensures the decontamination agent reaches all critical surfaces. Validation is essential for sterilization claims and regulatory defensibility.
Material, Equipment, and Process Controls
Materials and equipment can introduce contamination if not properly controlled.

Material introduction protocols
Controls may include:
- Staged wipe-downs.
- Double-bagging.
- Dedicating tools to rooms.
- Sterile packaging strategies.
Equipment cleanability and validation
- Surface compatibility.
- Cleaning method effectiveness.
Process segregation
CAPA, Investigations, and Continuous Improvement
No system is perfect. Deviations and excursions help facilities learn and strengthen their contamination control.
Root cause investigations
Investigations must examine:
- Human error.
- Equipment issues.
- Environmental conditions.
- Material problems.
Corrective and preventive actions
Effective CAPA includes:
- SOP updates.
- Retraining.
- Process redesign.
- Upgraded disinfectants or tools.
- Enhanced monitoring.
Ongoing review
Quality Review includes:
Regular CCS reviews, product quality reviews, and internal audit cycles help ensure the system evolves with operational needs.
The Role of Quality and Cross-Functional Alignment
QA ownership
- Risk assessments.
- SOP approvals.
- EM trend reviews.
- Deviation oversight.
- Validation alignment.
Cross-functional communication
Strong contamination control requires cooperation between:
- Operations.
- Cleaning teams.
- Engineering.
- Validation.
- QA.
- EM teams.
Culture of contamination control

Build a Stronger, Safer, Audit-Ready Contamination Control Program
Effective contamination control is not a single tactic—it is an integrated ecosystem that encompasses facility design, cleaning, disinfection, airflow, gowning, environmental monitoring, and quality oversight. With the right systems, personnel behaviors, and validated procedures, a facility can maintain consistent control, reduce risk, and protect both product and patient safety.
A well-structured contamination control program strengthens audit readiness, minimizes costly downtime, and supports uninterrupted manufacturing. If you need expert help evaluating your contamination prevention strategy, improving your cleaning program, or strengthening compliance practices, our team is ready to assist.